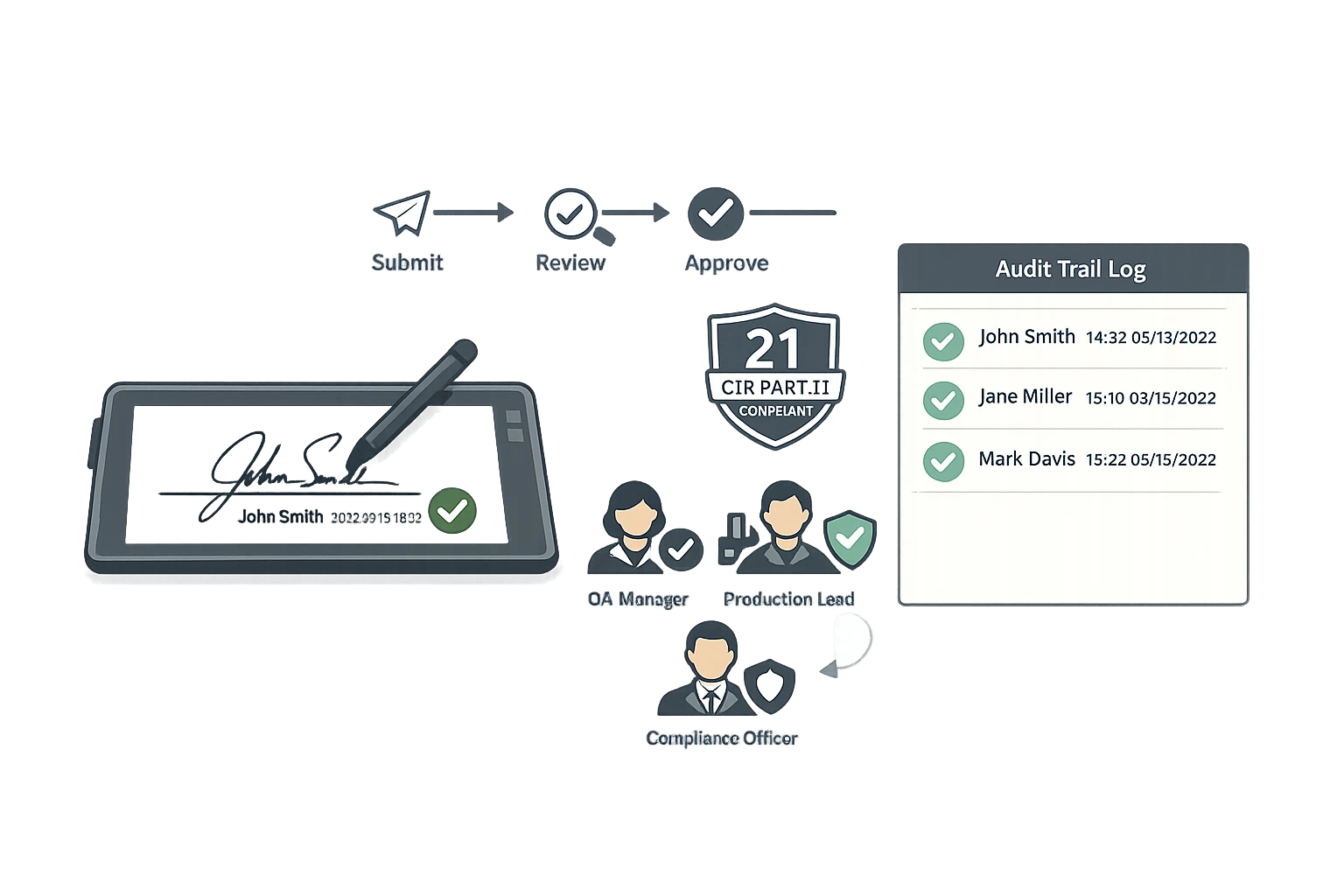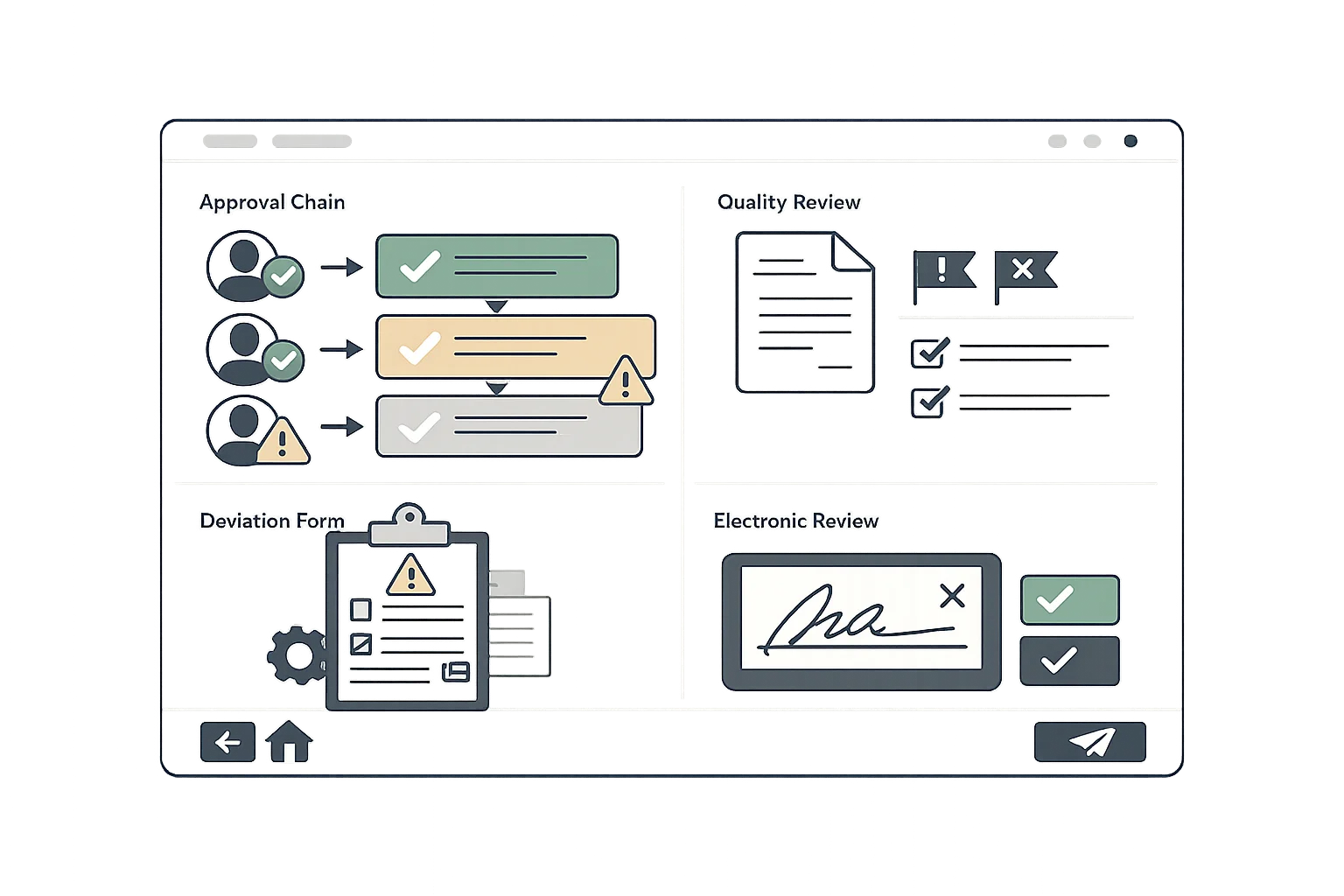
Electronic Production Records
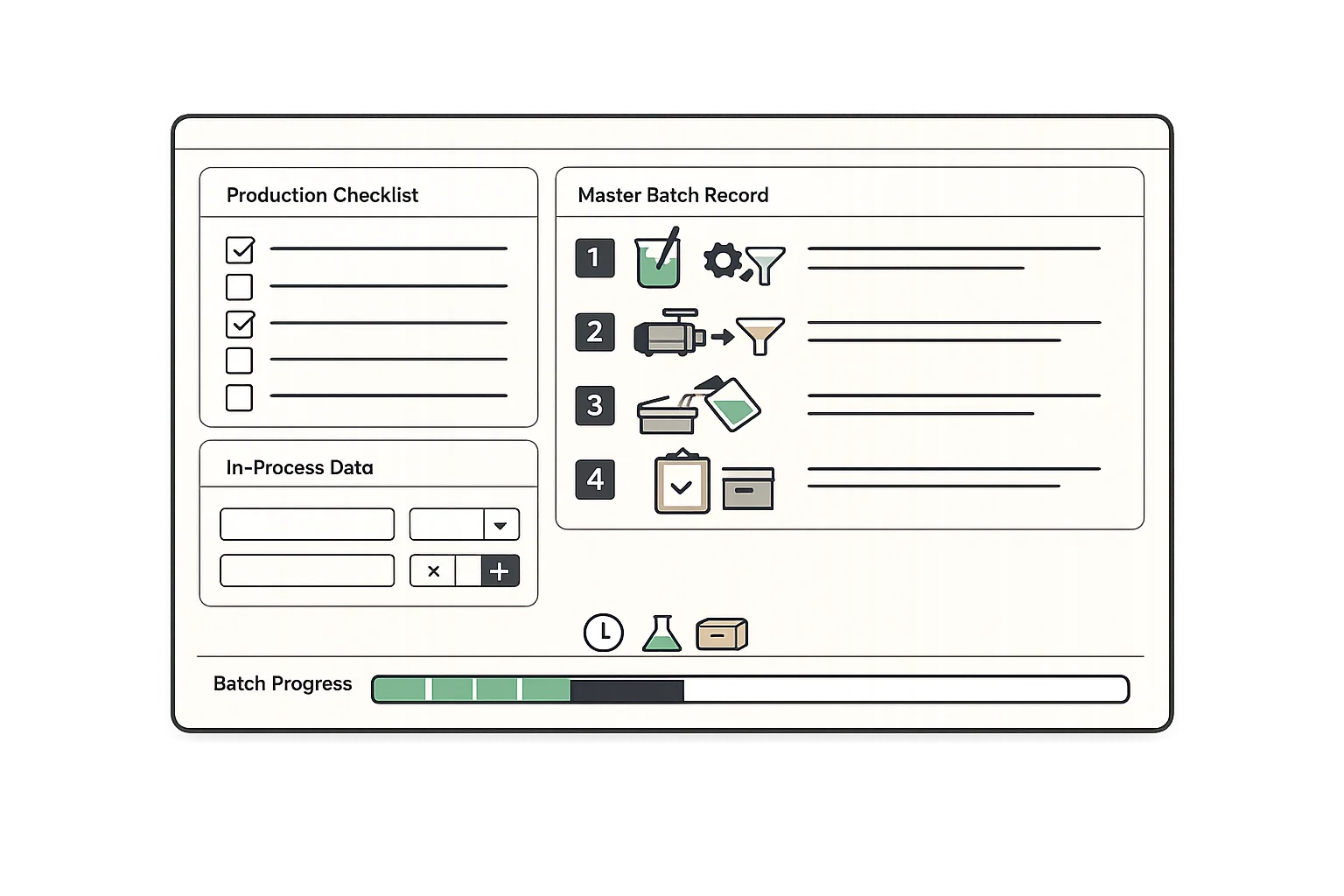
Replace paper production records with digital documentation that captures every production step. Ensure consistency across batches with master production record templates.
- Master production record templates
- Step-by-step production instructions
- In-process data capture
- Real-time batch progress tracking