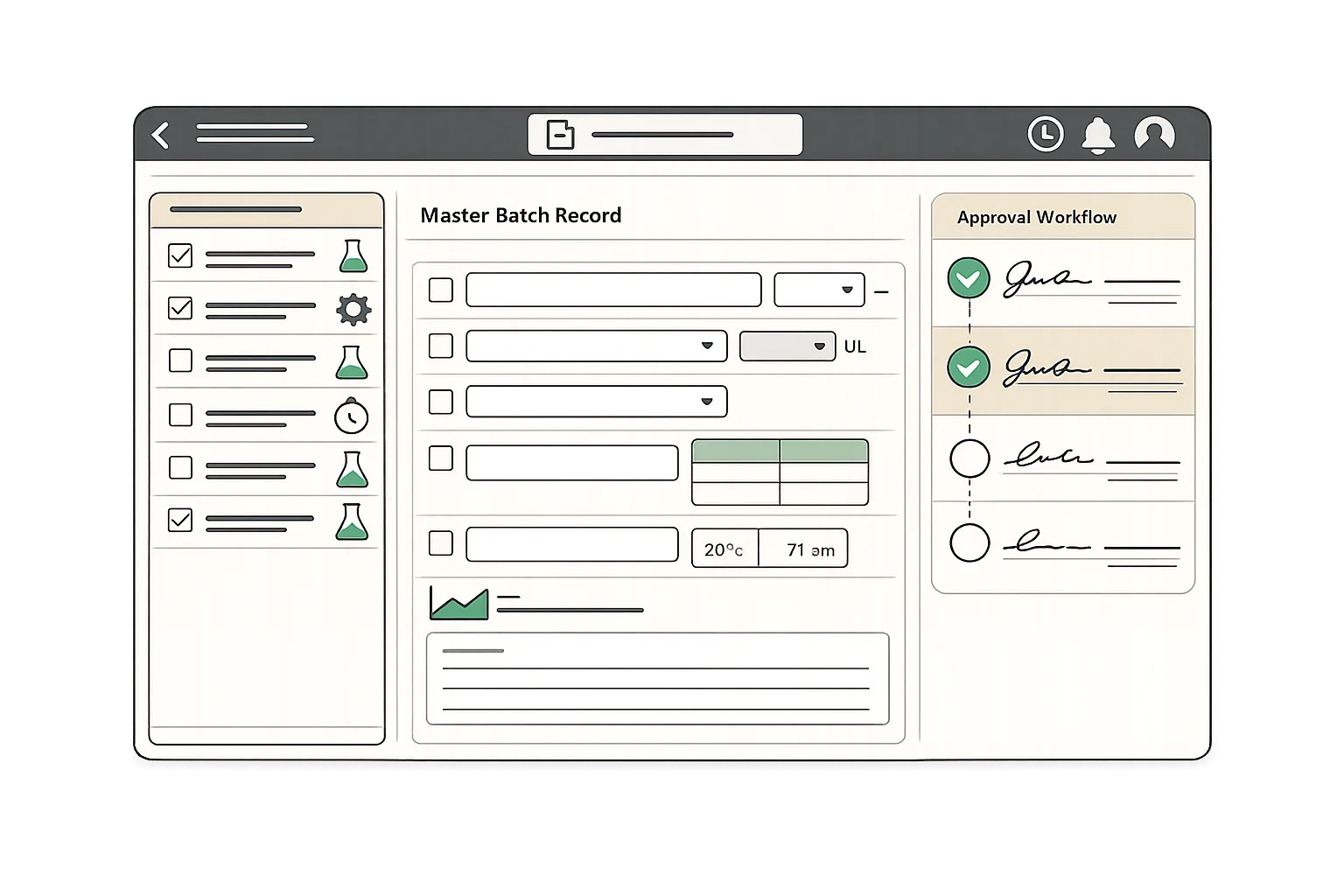
21 CFR Part 11 Electronic Records
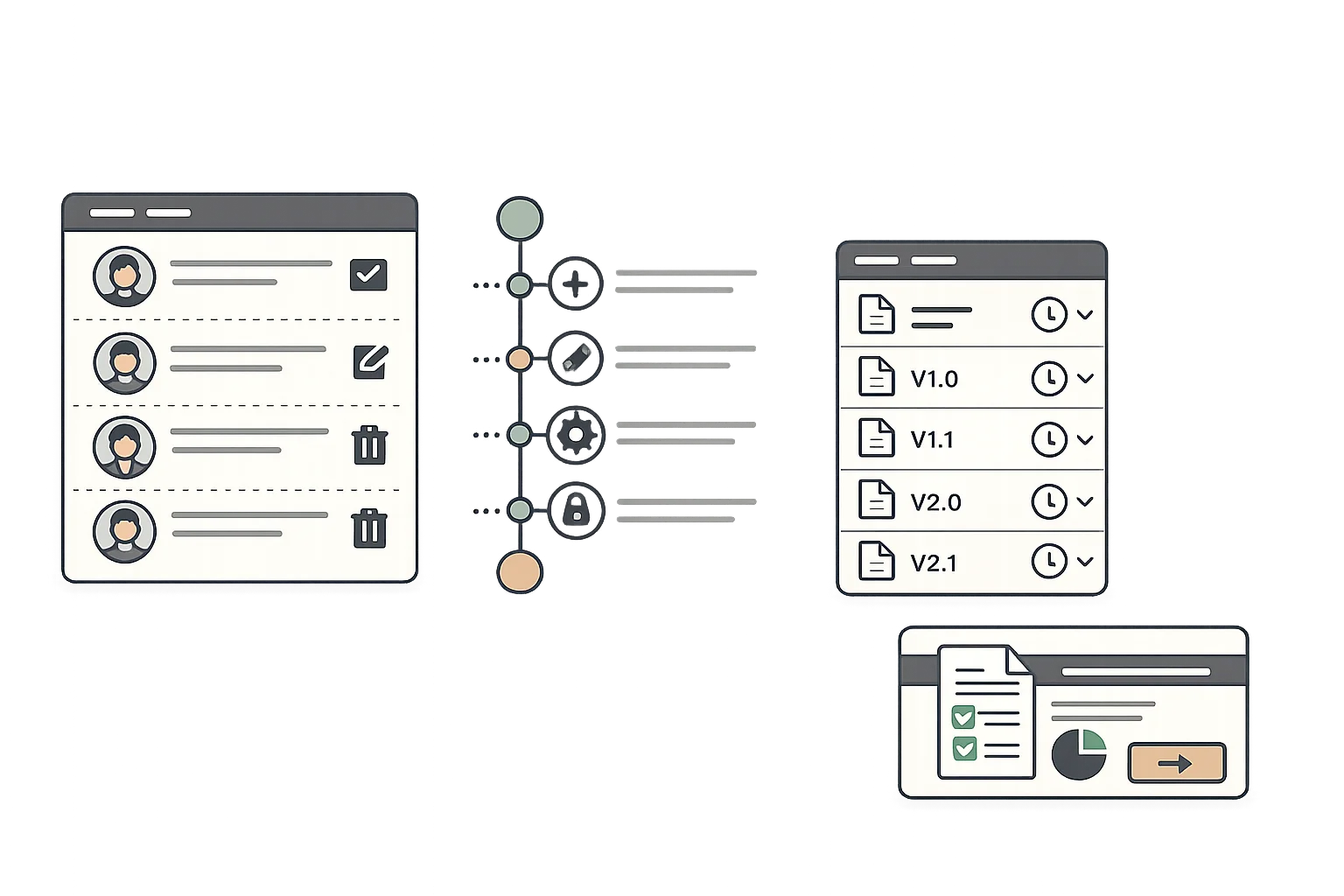
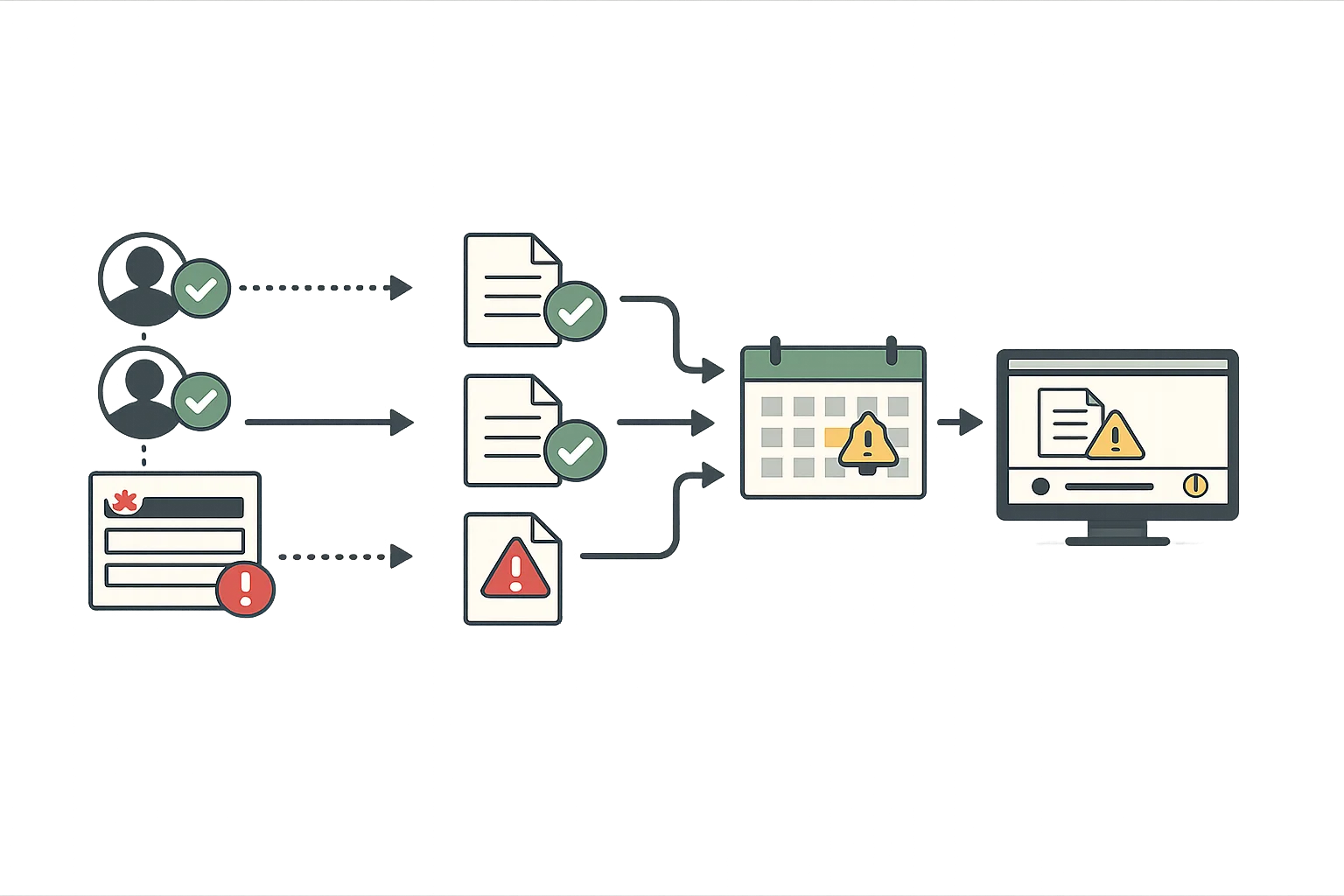
Meet FDA electronic record requirements with secure authentication, electronic signatures, and tamper-evident audit trails. Every record change is documented with user attribution and timestamps.
- Secure user authentication and access controls
- Electronic signatures with signer identification
- Complete audit trail for all record changes
- Tamper-evident record storage